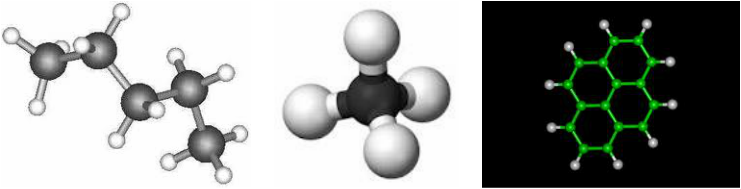
Hydrocarbons
- A hydrocarbon is a molecule made of just what is sounds like, Carbon and Hydrogen. The amont of Carbons determines the name.
- The majority of hydrocarbons found naturally occur in crude oil, where decomposed organic matter provides an abundance of carbon and hydrogen which, when bonded, can catenate to form seemingly limitless chains.
- Hydrocarbons are a primary energy source for current civilizations. The predominant use of hydrocarbons is as a combustible Fuel source.
Examples of Hydrocarbons
Some major hydrocarbons are Methane, Ethane, Propane, Butane, Pentane, Hexane, Heptane, Octane, Nonane, And Decane. Methane has 1 carbon and has a formula of CH4. Ethane has 2 Carbons and a formula of C2H6. Propane has 3 carbons and has a formula of C3H8. Butane has 4 carbons and a formula of C4H10. Pentane has 5 Carbon and a formula of C5H12. Hexane has 6 Carbons and a formula of C6H14. Heptane has 7 carbons and a formula of C7H16. Octane has 8 Carbons and a formula of C8H18. Nonane has 9 Carbons and a formula of C9H20. Decane has 10 carbons and a formula of C10H22.